Cusabio Human Recombinants
Recombinant Human Cementoblastoma-derived protein 1 (CEMP1) | CSB-YP764814HU
- SKU:
- CSB-YP764814HU
- Availability:
- 3 - 7 Working Days
Description
Recombinant Human Cementoblastoma-derived protein 1 (CEMP1) | CSB-YP764814HU | Cusabio
Alternative Name(s): Cementum protein 1Cementum protein 23 ;CP-23
Gene Names: CEMP1
Research Areas: Cell Biology
Organism: Homo sapiens (Human)
AA Sequence: MGTSSTDSQQAGHRRCSTSNTSAENLTCLSLPGSPGKTAPLPGPAQAGAGQPLPKGCAAVKAEVGIPAPHTSQEVRIHIRRLLSWAAPGACGLRSTPCALPQALPQARPCPGRWFFPGCSLPTGGAQTILSLWTWRHFLNWALQQREENSGRARRVPPVPRTAPVSKGEGSHPPQNSNGEKVKTITPDVGLHQSLTSDPTVAVLRAKRAPEAHPPRSCSGSLTARVCHMGVCQGQGDTEDGRMTLMG
Source: Yeast
Tag Info: N-terminal 6xHis-tagged
Expression Region: 1-247aa
Sequence Info: Full Length
MW: 28 kDa
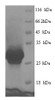
Purity: Greater than 90% as determined by SDS-PAGE.
Relevance:
Reference: Molecular cloning, expression and immunolocalization of a novel human cementum-derived protein (CP-23).Alvarez-Perez M.A., Narayanan S., Zeichner-David M., Rodriguez Carmona B., Arzate H.Bone 38:409-419(2006)
Storage: The shelf life is related to many factors, storage state, buffer ingredients, storage temperature and the stability of the protein itself. Generally, the shelf life of liquid form is 6 months at -20?/-80?. The shelf life of lyophilized form is 12 months at -20?/-80?.
Notes: Repeated freezing and thawing is not recommended. Store working aliquots at 4? for up to one week.
Function: May play a role in development of the periodontium which surrounds and supports the teeth by promoting the differentiation of multi-potent cells from the periodontal ligament into cementoblasts to form the cementum
Involvement in disease:
Subcellular Location: Cytoplasm, Nucleus
Protein Families:
Tissue Specificity: Expressed by cementoblasts, a subpopulation of periodontal ligament cells and cells located around vessels in periodontium (at protein level).
Paythway:
Form: Liquid or Lyophilized powder
Buffer: If the delivery form is liquid, the default storage buffer is Tris/PBS-based buffer, 5%-50% glycerol. If the delivery form is lyophilized powder, the buffer before lyophilization is Tris/PBS-based buffer, 6% Trehalose, pH 8.0.
Reconstitution: We recommend that this vial be briefly centrifuged prior to opening to bring the contents to the bottom. Please reconstitute protein in deionized sterile water to a concentration of 0.1-1.0 mg/mL.We recommend to add 5-50% of glycerol (final concentration) and aliquot for long-term storage at -20?/-80?. Our default final concentration of glycerol is 50%. Customers could use it as reference.
Uniprot ID: Q6PRD7
HGNC Database Link: HGNC
UniGene Database Link: UniGene
KEGG Database Link: KEGG
STRING Database Link: STRING
OMIM Database Link: OMIM